INTRODUCTION: T-cell malignancies are associated with frequent relapse and high morbidity in children and adults, partly due to the lack of effective or targeted treatment options. Despite the use of multidrug chemotherapy, <50% of adults and <75% of children with T-ALL survive longer than five years. While CAR-T cell therapy is remarkable in treating B-cell malignancies, its use in T-cell malignancies is restricted due to fratricide, risk of immunodeficiency, and immune escape.
METHODS: To broaden the use of CAR-T cells in pan T-cell malignancies, we developed an allogenic “universal” CD2-targeting CAR-T (UCART2), in which both CD2 and the T-cell receptor alpha subunit are both deleted by CRISPR/Cas9 editing to prevent both fratricide and GvHD. In vitro and in vivo efficacy of UCART2, alone or in combination with rhIL-7-hyFc (efineptakin alfa), a long-acting IL-7, was evaluated using CD2+ HH CBR-GFP T-ALL cell line and primary CD2+ T-ALL PDXs. To determine the impact of CD2 antigen loss on CAR-T cell function, CRISPR-mediated CD2 KO was performed in CD19 targeting CAR-T cells (UCART19ΔCD2). Single-cell secretome analyses on target-stimulated UCART19 and UCART19ΔCD2 were performed using the Isolight platform (IsoPlexis). The in vivo efficacy of UCART19 and UCART19ΔCD2 was evaluated in a CD19+ NALM6 GFP-CBR xenograft model.
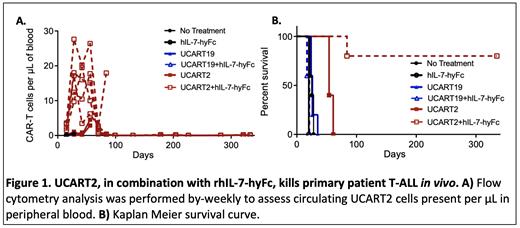
RESULTS: UCART2 demonstrated efficacy against T-ALL and CTCL in vitro and prolonged the survival of tumor-engrafted NSG mice in vivo. While CD2 deletion in UCART19 had little impact on in vitro killing efficacy, single-cell secretome analysis revealed that CD2 deletion in UCART19 reduced frequencies of the effector cytokines, including Granzyme-B and IFN-γ. We also observed that UCART19ΔCD2 (CD2-) had reduced anti-tumor efficacy compared to UCART19 (CD2+) in vivo in a CD19+ NALM6 GFP-CBR xenograft model. Of note, the reduced efficacy resulting from CD2 deletion was reversed when combined with rhIL-7-hyFc, a long-acting recombinant human interleukin-7. Treatment with rhIL-7-hyFc enhanced UCART2 expansion and persistence in vivo(Figure 1A) resulting in increased survival of NSG mice engrafted with both the primary patient T-ALL model (Figure 1B) and in a tumor re-challenge model in vivo.
CONCLUSIONS: We have developed an allogenic “universal” CD2-targeting CAR-T cell UCART2, which is effective against T-ALL and CTCL/T-NHL. We found that CD2 deletion in CAR-T cells resulted in reduced secretion of effector cytokines and reduced anti-tumor activity in vivo, indicating that CD2 is critical for CAR-T function. However, when combined with rhIL-7-hyFc (efineptakin alfa), a long-acting recombinant IL-7, UCART2 induced durable complete responses in both primary and tumor rechallenge experiments in vivo. Our data suggest that UCART2, in combination with rhIL-7-hyFc, could be a suitable approach for treating T-cell malignancies.
Disclosures
Carter:Wugen: Current equity holder in private company; BlueSphere Bio: Current Employment. Kim:NeoImmuneTech: Patents & Royalties. O'Neal:NeoImmuneTech: Patents & Royalties; Wugen: Patents & Royalties. Rettig:Integrated DNA Technologies: Current Employment. Turk:Integrated DNA Technologies: Current Employment. Wolfarth:NeoImmuneTech: Current Employment. Lee:NeoImmuneTech: Current Employment. Cooper:Wugen: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company, Patents & Royalties. DiPersio:Magenta: Other: Ownership Investment, Patents & Royalties; Vertex: Consultancy; Sun Pharma Ltd.: Membership on an entity's Board of Directors or advisory committees; hC Bioscience, Inc.: Membership on an entity's Board of Directors or advisory committees; Amphivena Therapeutics: Research Funding; RiverVest Venture Partners: Membership on an entity's Board of Directors or advisory committees; BiolineRx Ltd: Consultancy, Research Funding; Incyte: Consultancy, Research Funding; NeoImmune Tech: Consultancy; Macrogenics: Consultancy, Research Funding; Washington University: Current Employment; WUGEN: Other: Ownership Investment, Patents & Royalties.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal